chadwick's discovery of the neutron
James Chadwick was educated at the universities of Manchester, Berlin, and Cambridge. At Manchester Chadwick actually studied under Ernest Rutherford. Chadwick eventually took over Rutherford's research and studies a bit later in his life. Chadwick's discovery of the neutron marked the beginning of the modern era of study and understanding of the atomic nucleus.
Before the discovery of the neutron, atoms were thought to have been made of two types of particles: protons and electrons. The protons were bunched up in an atom's nucleus and the electrons just orbited the nucleus. Earlier experiments shows that the nucleus in a helium atom gave off exactly two times as much electrical force on an electrons as a nucleus in a hydrogen atom. The only problem with this, was that helium would then have two protons and two electrons, making it's mass twice that of a hydrogen atom, which has one proton and one electron. The reason that this was an issue was because in reality, helium has a mass four times that of hydrogen. Chadwick hypothesized that a helium nucleus had two more particles that were not discovered yet. He also thought that these two additional particles didn't have a charge, because he knew that they weren't attracted in any way to any sort of electrical force. Many other scientists thougth that the particles that Chadwick was talking about were only gamma rays, but gamma rays don't have any mass, so it wouldn't make sense. Therefore, Chadwick decided to try and find out the new particle's mass. This was very difficult however, because a subatomic particle can not just be put on a scale and weighed. But Chadwick came up with a solution.
Many nuclei of different chemical elements had masses that were already known, and some even already had special ways that you could measure the speed of a quickly moving nucleus. Chadwick then set out to strike sampled of certain elements with the unknown new particles. When there was a direct impact between the unknown particle and the nucleus of one of the atoms, the nucleus would be hit out of the atom, and then Chadwick would record its speed.
Before the discovery of the neutron, atoms were thought to have been made of two types of particles: protons and electrons. The protons were bunched up in an atom's nucleus and the electrons just orbited the nucleus. Earlier experiments shows that the nucleus in a helium atom gave off exactly two times as much electrical force on an electrons as a nucleus in a hydrogen atom. The only problem with this, was that helium would then have two protons and two electrons, making it's mass twice that of a hydrogen atom, which has one proton and one electron. The reason that this was an issue was because in reality, helium has a mass four times that of hydrogen. Chadwick hypothesized that a helium nucleus had two more particles that were not discovered yet. He also thought that these two additional particles didn't have a charge, because he knew that they weren't attracted in any way to any sort of electrical force. Many other scientists thougth that the particles that Chadwick was talking about were only gamma rays, but gamma rays don't have any mass, so it wouldn't make sense. Therefore, Chadwick decided to try and find out the new particle's mass. This was very difficult however, because a subatomic particle can not just be put on a scale and weighed. But Chadwick came up with a solution.
Many nuclei of different chemical elements had masses that were already known, and some even already had special ways that you could measure the speed of a quickly moving nucleus. Chadwick then set out to strike sampled of certain elements with the unknown new particles. When there was a direct impact between the unknown particle and the nucleus of one of the atoms, the nucleus would be hit out of the atom, and then Chadwick would record its speed.
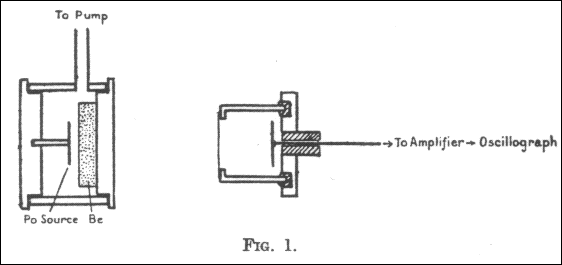
In order to be able to conduct this experiment, Chadwick had to construct a subatomic pool table. In this apparatus, there is a disk of polonium. Polonium gives a source of radiation which makes it possible to hit the neutrons out ot the beryllium nuclei. The radiation given off by the polonium is absorbed by a few millimeters of air, and so that air must be pumped out of the left-hand chamber. The unkown particles, now known as neutrons, strike matter more readily, and they shoot out through the wall and right into the right-side chamber, which is full of both hydrogen and nitrogen gas. When a neutrons strikes the hydrogen or nitrogen nucleus, it shoot right out of the atom with lots of force, this nucleus then splits into thousand of other atoms of the gas. The result is an electrical pulse that is recorded by the wire located on the right of the diagram. Physicists were then able to take the strength of the electrical pulse and make it into a measure of force. Chadwick conducted another experiment just like this one but he used nitrogen instead. With the information from these two experiments, Chadwick was then able to solve for the mass of the unknown particle. The particle's mass was less than 1% than that of the mass of a proton. Since the new particle was electrically neutral, he named it the nuetron.