Quantum Numbers in Electron Configuration
http://cdn.dipity.com/uploads/events/a16771e509158b5df1b0893f30d932b0_1M.png
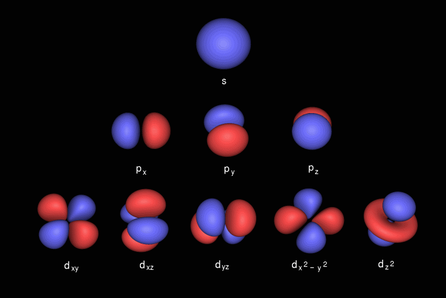
Quantum numbers main purpose is to explain where any electron is positioned in an atom and moreover how all the electrons in an atom are positioned in respect to one another. For information on the nature of quantum numbers and what the four different quantum numbers are go here http://nuclearchem101.weebly.com/quantum-numbers.html. When it comes to the role of quantum numbers in electron configuration their role is simple. The principle quantum number stipulates the electron shell that a particular electron is in. The angular quantum number specifies the subshell that you are in for your particular shell (to the right you can see the electron orbitals possible in the first 3 subshells). The magnetic quantum number describes which orbital you are in in you particular shell, as you can see in the image above some subshells have different possible orbitals that have the same energy (this is why they end up in the same subshell). The magnetic quantum number is so important because it explains why a subshell like the P subshell can contain 6 electrons in total when every cloud can only hold two electrons. It is because the magnet quantum number allows for multiple clouds in the same subshell. The final quantum number is the spin quantum number, which allows for every electron cloud to contain two electrons and yet still maintain adherence to the Pauli Principle. The spin quantum number is either 1/2 or -1/2 one denoting clockwise spin-like behavior and the other counterclockwise.
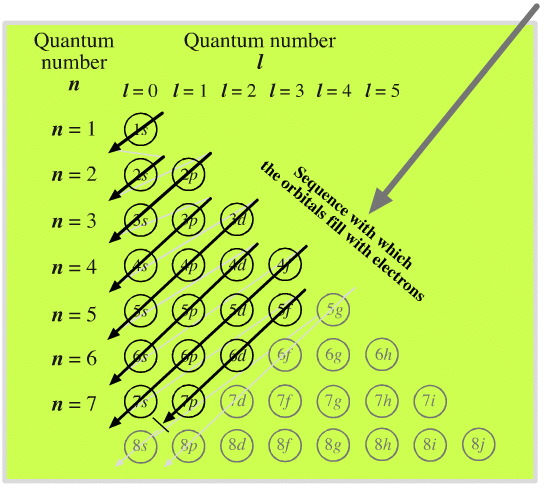
Although the angular quantum number itself can have any integral value, that isn't necessarily true in the grand scheme of things. Eventually if certain quantum numbers have certain values they can begin to contradict other rules. Sometimes they don't contradict rules but instead they are merely theoretical. This useful chart below can help you figure out which quantum numbers and subshells actually pertain to the periodic table.
Although the angular quantum number itself can have any integral value, that isn't necessarily true in the grand scheme of things. Eventually if certain quantum numbers have certain values they can begin to contradict other rules. Sometimes they don't contradict rules but instead they are merely theoretical. This useful chart below can help you figure out which quantum numbers and subshells actually pertain to the periodic table.
Since all the subshells after the F subshell are theoretical, you rarely ever see an l quantum number greater than 3 in electron configuration. The same thing can be said of the n quantum number as anything over n=7 is purely theoretical.
In summation, electrons configure in the pattern and order that they do into subshells because of the laws governing quantum numbers. These quantum numbers also determine the location of the probability cloud than an electron inhabits and how the different clouds are positioned in relation to one another to describe their electron location on a whole.
In summation, electrons configure in the pattern and order that they do into subshells because of the laws governing quantum numbers. These quantum numbers also determine the location of the probability cloud than an electron inhabits and how the different clouds are positioned in relation to one another to describe their electron location on a whole.