Rutheford's Experiment - Proving the Existence of a Nucleus
http://www.daviddarling.info/images/Rutherford_gold-foil_experiment.jpg
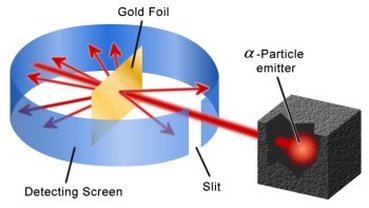
Using his gold foil experiment, he was able to prove the existence of the atomic nucleus and steer the world of science on the right course in terms of the nuclear model, pioneering the Rutheford model after his famous gold foil experiment. At the time of this gold foil experiment the generally accepted atomic model was the plum pudding model developed by J. J. Thompson who had discovered the electron some years earlier. The purpose of the experiment was essentially to prove Thompson's theorized model right. An alpha emitter would be placed in a prohibitive box where alpha particles could only escape through a small hole. From there they would travel through the slit in the detection screen diagrammed above and come in contact with a piece of gold foil only a single atom thick. Since the plum pudding model stated that each atom was a positively charged pudding speckled with electrons (plums), he expected the alpha particles to pass straight through the gold foil or to be deflected slightly if they came in contact with an electron. However, as the experiment panned out the angles of particle deflection were much greater than theorized and some were even deflected straight back at the alpha emitter. This ultimately proved the plum pudding model incorrect and gave rise to Rutheford's explanation for the experiment, the nucleus.
