the nucleus
http://4.bp.blogspot.com/-R5eP8hz_bBM/TiBCidsz1lI/AAAAAAAAABI/U2QkhjLdNC8/s1600/atom.JPG
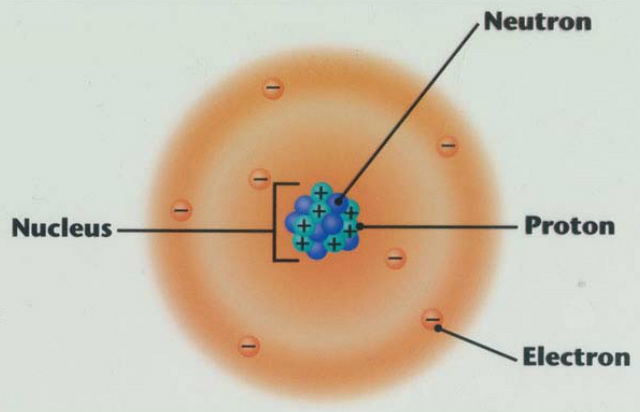
What is a nucleus of an atom?
-Well, the nucleus of an atom is the very center of the atom.
What does the nucleus consist of?
-The nucleus of an atom consists of two types of subatomic particles: protons, which are positively charged, and neutrons, which are neutrally charged. However, electrons play a role in the atom, even though they are located outside the nucleus.
But how does the nucleus stay together? What keeps electrons inside the atom?
-There are two main things that keep the nucleus together; electric force and strong force. With electric force, there are protons, which have a positive charge, and electrons which have a negative charge. Every proton has the exact same charge, and every electron has the exact opposite charge of a proton. Therefore, when two protons repel each other in the nucleus, there repelling is basically counteracted by the charge of the electron. Now for strong force, it is a force which attracts protons to protons, neutrons to neutrons, and protons to neutrons. So in an atom, its nucleus is held together by strong force, and the electrons outside are held in the atom by the electric force.
-Well, the nucleus of an atom is the very center of the atom.
What does the nucleus consist of?
-The nucleus of an atom consists of two types of subatomic particles: protons, which are positively charged, and neutrons, which are neutrally charged. However, electrons play a role in the atom, even though they are located outside the nucleus.
But how does the nucleus stay together? What keeps electrons inside the atom?
-There are two main things that keep the nucleus together; electric force and strong force. With electric force, there are protons, which have a positive charge, and electrons which have a negative charge. Every proton has the exact same charge, and every electron has the exact opposite charge of a proton. Therefore, when two protons repel each other in the nucleus, there repelling is basically counteracted by the charge of the electron. Now for strong force, it is a force which attracts protons to protons, neutrons to neutrons, and protons to neutrons. So in an atom, its nucleus is held together by strong force, and the electrons outside are held in the atom by the electric force.
