Thomson Cathode ray tube experiment
http://images.tutorvista.com/content/feed/u2242/cathode.JPG
Electrons were identified by the English physicist Joseph J. Thomson in 1897. The term "cathode ray" was first used to describe the particles that are now called electrons. Thomson used a cathode-ray tube to make cathode rays, which are negatively charged particles that are made by a cathode, a substance that emmits electrons. Thomson's research proved the existence of negatively charged particles, now called electrons. Thomson actually won a Nobel Prize for physics. Thomson's research and discoveries led to later experiments by Bohr and Rutherford, leading to an understanding of the structure of an atom.
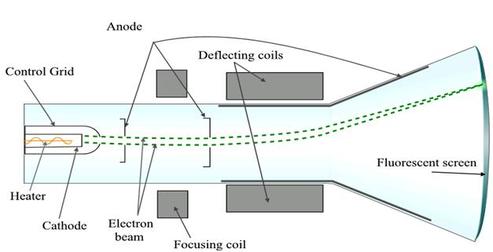
Thomson had three cathode-ray tube experiments. For the first one of his experiments, Thomson tried to prove that the rays emitted from the cathode-ray tube were inseperable from the latent charge. For this experiment, he used a magnetic field. Thomson built a cathode-ray tube, with a metal cylinder on the end. The cylinder on the end of the cathode-ray tube had two small cuts which had electrometers that could measure electric charges that were very small. Thomson discovered that when he put a magnetic field across the tube, there weren't any electric charges measured by the electrometers on the cathode-ray tube. From the results of his first experiment, Thomson found out that the cathode ray and the negative charge were not able to separated. Thomson continued his experimenting and conducted a second experiment, in which he aimed to prove that the cathode rays were negatively charged. In his experiment, he tried to deflect the rays with an electric field. He made a cathode-ray tube that was different from others. For this cathode-ray tube, Thomson coated one end with fluorescent coating and a vacuum. He put two electric plates towards the middle of the tube. Those electric plates caused the making of a negative cathode and a positive anode. Thomson used the negative cathode and positive anode to try and deflect the cathode rays. As Thomson had hypothesized, the cathode rays were deflected by the electric charge. This further provided evidence that negatively charged particles made up the cathode rays. Thomson went on to perform a third experiment, in which he attempted to find out the nature of the particles. The particles were very small, so small that they were too small in order to find out their charge or mass. However, Thomson tried to accomplish this by recording how much the particles were bent by electrical currents that had different forces. Thomson discovered that the ratio of charge to mass was incredibly large, and that the particles either had to be nearly a thousand times smaller than a hydrogen ion or they had to have an incredible charge. Thomson then made a big step, saying that the cathode rays were made of particles that emanated from within the atoms themselves.
Thomson came up with the first real understanding of an atom's structure, and how it had negatively charged particles around it. Thomson's discoveries led to many other discoveries in quantum physics, especially since Thomson found out how atom's had complex internal structures. Thomson's findings also helped later physicists and their discoveries. Thomson's findings from his experiments helped later scientists make revolutionary discoveries but it was all possible because of Thomson's cathode-ray tube experiments.
Thomson had three cathode-ray tube experiments. For the first one of his experiments, Thomson tried to prove that the rays emitted from the cathode-ray tube were inseperable from the latent charge. For this experiment, he used a magnetic field. Thomson built a cathode-ray tube, with a metal cylinder on the end. The cylinder on the end of the cathode-ray tube had two small cuts which had electrometers that could measure electric charges that were very small. Thomson discovered that when he put a magnetic field across the tube, there weren't any electric charges measured by the electrometers on the cathode-ray tube. From the results of his first experiment, Thomson found out that the cathode ray and the negative charge were not able to separated. Thomson continued his experimenting and conducted a second experiment, in which he aimed to prove that the cathode rays were negatively charged. In his experiment, he tried to deflect the rays with an electric field. He made a cathode-ray tube that was different from others. For this cathode-ray tube, Thomson coated one end with fluorescent coating and a vacuum. He put two electric plates towards the middle of the tube. Those electric plates caused the making of a negative cathode and a positive anode. Thomson used the negative cathode and positive anode to try and deflect the cathode rays. As Thomson had hypothesized, the cathode rays were deflected by the electric charge. This further provided evidence that negatively charged particles made up the cathode rays. Thomson went on to perform a third experiment, in which he attempted to find out the nature of the particles. The particles were very small, so small that they were too small in order to find out their charge or mass. However, Thomson tried to accomplish this by recording how much the particles were bent by electrical currents that had different forces. Thomson discovered that the ratio of charge to mass was incredibly large, and that the particles either had to be nearly a thousand times smaller than a hydrogen ion or they had to have an incredible charge. Thomson then made a big step, saying that the cathode rays were made of particles that emanated from within the atoms themselves.
Thomson came up with the first real understanding of an atom's structure, and how it had negatively charged particles around it. Thomson's discoveries led to many other discoveries in quantum physics, especially since Thomson found out how atom's had complex internal structures. Thomson's findings also helped later physicists and their discoveries. Thomson's findings from his experiments helped later scientists make revolutionary discoveries but it was all possible because of Thomson's cathode-ray tube experiments.
