periodic trends Dmitri Mendeleev, a Russian Chemist came up with the current element arrangement in the periodic table. But how did he arrange them? What properties of elements did he use to arrange them into the table? What patterns are there in the table?
|
http://en.wikipedia.org/wiki/File:Periodic_trends.svg
- Groups and Periods
-In the periodic table there are 18 groups and 7 periods. Groups 1A-12A, and 3B-8B, and periods 1-7. Whatever period an element is in is how many shells it has in its electron configuration. For example, since Rubidium is period 5, Rubidium will have 5 shells when you draw out its electron configuration.
-With the exception of transition metals, Lanthanoids, and Actinoids, whatever group an element is in determines how many valence electrons that is has. For example, Nitrogen is in group 5B, so it has 5 electrons.
-Atomic Radius
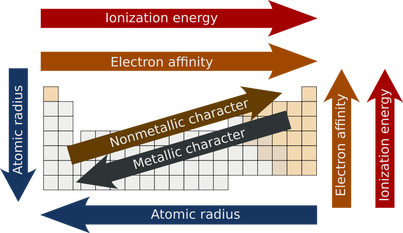
-The radius of an atom increases as you go down the table. An element in period 1 will have an atomic radius smaller than an element in period 7. Also, as you move across the table, the radius of an atom decreases.
-Electron Affinity (the energy released when a neutral atom as a gas gets another electron to make an anion)
-The lower down you go in a group, the more the electron affinity decreases.
-As you go right in a period, the more the electron affinity increases.
-But that's not it, of course there are exceptions:
The elements in the nonmetals in the first period have electron affinities that are actually lower than the elements below them in their groups. Also, if an element has an electron configuration of Xs2, Xp3, and Xp3, it will have an electron affinity that is negative. This is, because they are for the most part unstable.
-First Ionization Energy (the energy needed to release the outermost electron out of a neutral atom in its natural state)
-First ionization decreases as you go down a group. This is, because the outermost electron is much easier to get rid of because the electrons are farther away from the nucleus, and not as strongly attracted.
-First ionization increases as you go right in a period. This is, because as you go right in a period the atoms are smaller, and the valence electrons are closer to the nucleus, so they need more energy to remove them, because they have a stronger attraction to the nucleus.
-In the periodic table there are 18 groups and 7 periods. Groups 1A-12A, and 3B-8B, and periods 1-7. Whatever period an element is in is how many shells it has in its electron configuration. For example, since Rubidium is period 5, Rubidium will have 5 shells when you draw out its electron configuration.
-With the exception of transition metals, Lanthanoids, and Actinoids, whatever group an element is in determines how many valence electrons that is has. For example, Nitrogen is in group 5B, so it has 5 electrons.
-Atomic Radius
-The radius of an atom increases as you go down the table. An element in period 1 will have an atomic radius smaller than an element in period 7. Also, as you move across the table, the radius of an atom decreases.
-Electron Affinity (the energy released when a neutral atom as a gas gets another electron to make an anion)
-The lower down you go in a group, the more the electron affinity decreases.
-As you go right in a period, the more the electron affinity increases.
-But that's not it, of course there are exceptions:
The elements in the nonmetals in the first period have electron affinities that are actually lower than the elements below them in their groups. Also, if an element has an electron configuration of Xs2, Xp3, and Xp3, it will have an electron affinity that is negative. This is, because they are for the most part unstable.
-First Ionization Energy (the energy needed to release the outermost electron out of a neutral atom in its natural state)
-First ionization decreases as you go down a group. This is, because the outermost electron is much easier to get rid of because the electrons are farther away from the nucleus, and not as strongly attracted.
-First ionization increases as you go right in a period. This is, because as you go right in a period the atoms are smaller, and the valence electrons are closer to the nucleus, so they need more energy to remove them, because they have a stronger attraction to the nucleus.
