The Millikan Oil Drop Experiment: Determining the Charge on an Electron
http://ffden-2.phys.uaf.edu/212_fall2003.web.dir/ryan_mcallister/slide3.htm
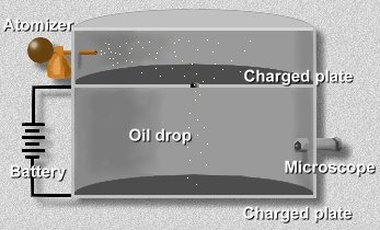
In 1909, Robert Millikan found out the size of a charge on an electron, as well as the fact that there was a smallest unit charge. For this work, he actually received a Nobel Prize. In his experiment, Millikan was able to find out the size of a charge on an electron. For his experiment, Millikan put a charge on a small drop of oil. He then recorded how strong an electric field had to be to stop the oil drop from falling. Millikan was able to find the mass of the drop, so he could find out the gravitational force on the one oil drop. He then was able to find the electric charge that drop needed to have. After he used different charges on different drops, he was able to determine that the charge was a multiple of -1.6 x 10^-16 coulombs (coulombs being the international system of units for measuring electric charge) . What this meant was that the electrons were carrying the unit charge. For this experiment, Millikan used an apparatus (a diagram of the apparatus used in his experiment is shown above). In this apparatus, an atomizer, a device that emits water or other liquids as a fine spray, sprayed a fine mist of the oil drops into the chamber. Some of the oil drops fell through a hole in the upper part of the apparatus. Millikan let the oil droplets fall, up to point when they reached terminal velocity (the maximum acceleration of something falling vertically towards Earth). Using the microscope in the apparatus, Millikan measured the terminal velocity, and by using a special formula, Millikan was able to find out the mass of each oil drop. Afterwards, he put a charge on the falling oil droplets by using x-rays to irradiate the lower chamber. This made the air become ionized, because the atoms in the air were converted into ions by removing one or more electrons. Because the air was ionized, electrons attached themselves to the oil drops, since he had previously put a charge on the droplets. He then put a battery on the plates that were located above and below the lower chamber, allowing him to apply an electric voltage. This electric field that was made in the lower chamber affected the charged oil drops. If it was set to a certain voltage, then the electromagnetic force produced would balance the gravitational force on a drop, and then the drop would stay suspended in mid-air.
In this experiment, the electromagnetic force pushed the oil droplets up, and gravity is what pulled the droplets down. Millikan used this data to determine the charge on a single electron using the behavior of the oil drop. He found that the charge on an electron was a multiple of -1.6 x 10^-16 coulombs.
In this experiment, the electromagnetic force pushed the oil droplets up, and gravity is what pulled the droplets down. Millikan used this data to determine the charge on a single electron using the behavior of the oil drop. He found that the charge on an electron was a multiple of -1.6 x 10^-16 coulombs.
