The Uncertainty Principle
http://www.satmagazine.com/cgi-bin/display_image.cgi?1454402056
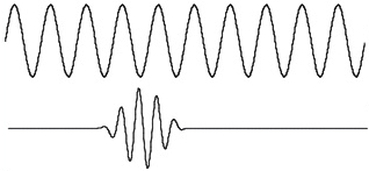
The uncertainty principle is one of the key principles that explains the quantum mechanical world. The concept was discovered by Werner Heisenberg, a german physicist who was a pioneer in the field of quantum mechanics. The Uncertainty Principle states that there is a limit to how much you can know about a particle's position and momentum, two fundamental properties of a particle, at the same time. In the most simple sense the uncertainty principle states that the more you know about where a particle is the the less you know about how fast it's going. In reality it gets quite a bit more complicated than that. The Uncertainty Principle can be explained with the wave-particle duality that exists in Quantum Mechanics. Since a particle behaves both like a wave and a particle at the same time, we can look to the behavior of waves to explain the uncertainty principle's effect on particles. When measuring a wave's momentum or frequency, you need to look at the wave on a whole like the top wave displayed to the left. However, when looking at a wave on the whole you cant actually identify its exact position. To determine the exact position of a wave you have to look at a wave pulse similar to the one depicted in the bottom curve in the image to the left. In effect, when you are able to precisely identify a wave's momentum you dont know much about its location and vice versa.
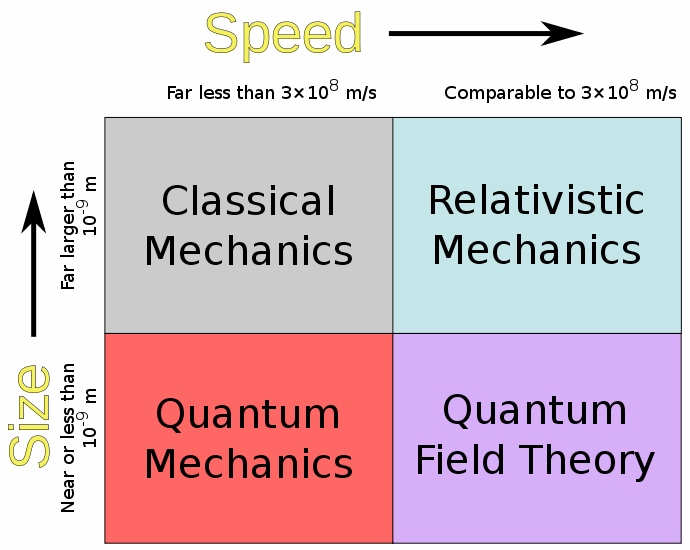
This has a tremendous effect on the very nature of Quantum Mechanics and is one of the key differences between Classical and Quantum Mechanics. Classical Mechanics lies in the realm of Newtonian Physics; that is it is only applicable to systems that have a certain size and speed both depicted in the image above as well as being based off of Isaac Newton's laws of motion. In classical mechanics, there is always certainty when it comes to measurements. There are steadfast values for everything. When you go down onto the micro-scale into the realm of Quantum Mechanics, this idea falls apart. Quantum Mechanics is an entirely new set of rules for the physical world that only applies under certain conditions. Most of Quantum Mechanics is probabilistic and one of the things that the uncertainty principle has the largest effect on is probabilities. A good example of this in action are the electron orbitals; the probabilities used to create the orbits are all governed by the uncertainty principle. In fact all of the quantum universe obeys the uncertainty principle, although it may not be obvious at first.